Start-Up
Whether a new intervention or program will be small or large, at a single site, or planned for widespread implementation after startup, there are several domains to consider when building the foundation of a program. This Section highlights these key domains based on our experiences with the Zika Contraception Access Network (Z-CAN) program design and startup in Puerto Rico. Some considerations, such as procurement and supply chain for contraceptive methods, are specific to a contraception intervention. Others, such as data collection and management, may be adapted to any service intervention or program.
The domains considered in this Section are not all encompassing for all possible interventions or programs; therefore, depending on the program, there may be other domains not addressed here that may be essential to the success of the proposed intervention or program. This Section includes guidance on domains included in Z-CAN program start-up.
Recruiting health care providers is the first step to building capacity among a network of health care providers who will implement the program. The process does not have to be lengthy, but recruitment does require careful consideration. You may need to tailor these steps to fit your specific context, culture, and service delivery norms.

Recruitment checklist topic: What types of health care providers are providing services in the target area?
Once you have selected to pool of health care providers that is the best fit for the program intervention and training activity, assess their baseline knowledge and competency through informal discussions or a short quantitative assessment with select health care providers. This step will clarify gaps in skills and help with the development of a targeted training. It can also influence the level of onsite mentorship that may be needed following training.
RESOURCE AVAILABLE:
Z-CAN Example: Provider Pool
In Puerto Rico, contraception activities are provided by obstetrics and gynecology (ob-gyn) physicians. Puerto Rico does not use certified registered nurse practitioners, physician assistants, clinical nurse specialists, certified registered nurse anesthetists, or certified nurse midwives for the provision of contraceptive services. Other primary care physicians often are restricted by insurance coverage from providing services such as long-acting reversible contraception (LARC). For these reasons, the Z-CAN provider network was almost exclusively ob-gyn physicians. Through the needs assessment we found there was limited provision of LARC methods in Puerto Rico prior to Z-CAN, which indicated a need to provide intrauterine device (IUD) and implant insertion and removal simulation training and onsite mentorship.

Checklist topic: What policies govern your setting?
Be sure you understand the national, regional, or facility-based policies that govern your setting. These policies may guide the scope of practice, clinical norms, and access to care. Understanding these polices will help in efficient and effective recruitment of health care providers. Even if polices are restrictive, it is important to respect and work within the existing policies. If a change in a policy could improve access to contraception services or other services of interest, the program can work with key stakeholders and policymakers to generate an evidence base that may support eventual policy change.
Lessons Learned: Recruitment and Training Policy
Understand and respect any supervisory hierarchy. Some health care providers may not be able to participate in a training activity and program unless they are recruited through their supervisor or have their supervisor’s permission to participate.

Checklist topic: Who can help access health care providers from the desired pool of providers?
Find leaders in local professional societies, the public health system and relevant divisions within that system, or other provider organizations. If you will need facility and clinic staff other than the health care providers you will target to provide the new intervention, participating health care providers can recruit from their home sites using eligibility criteria provided by the program. A clear statement of the activity should accompany the notification of and recruitment for training.
Z-CAN Example: Provider Recruitment
Program staff consulted with a wide range of agencies and organizations to recruit health care providers for Z-CAN. We worked with the Puerto Rico Department of Health (PRDOH) to identify ob-gyns on the island and with the local section of the American College of Obstetricians and Gynecologists (ACOG) to recruit private physicians. Consultation with the Medicaid program identified ob-gyns who practice within the Medicaid Managed Care Organizations. The Puerto Rico Primary Care Association helped connect Z-CAN with ob-gyns who practice in community health centers, and the University of Puerto Rico helped Z-CAN connect with residents in the medical schools.

Checklist topic: What is the best way to contact and recruit health care providers in my setting?
Understand the norms of the specific context in selecting your method of recruitment. Recruiting well in advance of a training will allow time for health care providers to get necessary approvals, book time away from work, and generate maximum participation.
A Physician Agreement that establishes a formal relationship between the physician and a program and the agency or organization overseeing the program intervention is an essential safeguard for both parties delivering a program intervention. This agreement typically defines the scope of work, obligations, terms and conditions, confidentiality requirements, and other legal parameters. Payment to health care providers, systems, and facilities varies across countries, states, and communities; therefore, the Physician Agreement should adhere to the laws and practices in your setting. For example, a physician working under contract with a Ministry of Health might not be eligible to receive outside support or income. In contrast, a private physician could be reimbursed for participating in a program. It is recommended that program development staff seek guidance from counsel to develop a comprehensive agreement tailored to the program needs and ensure compliance to local laws and regulations.
Programmatic expectations for the physician and medical practice staff can be further clarified by developing a provider manual that establishes policies and procedures for the program. The manual can include detailed reports and forms the physician’s medical practice must complete and specify how and when to submit these items.
Z-CAN Example: Physician Agreement
Physicians participating in the Z-CAN program signed a Physician Agreement with the National Foundation for the Centers for Disease Control and Prevention, Inc. (CDC Foundation) to provide contraceptive services and products to women at no cost. The agreement established reimbursement fees to physicians in private practice based on average Medicaid reimbursement rates in the continental United States. Health Resources and Services Administration (HRSA)-funded community health center clinics were also part of the Z-CAN network and provided Z-CAN services and methods at no cost to patients. An agreement established between the CDC Foundation and the HRSA community health center clinics provided Z-CAN-trained physicians employed by those clinics access to the full range of reversible contraceptive methods at no cost. However, the Z-CAN program did not provide physician reimbursement at community health centers because the HRSA prospective payment system used by community health centers was considered adequate.

Some of the key components of the Z-CAN Physician Agreements included:
- Detailed scope of work
- Record-keeping requirements
- Indemnification provisions
- Confidentiality provisions
- Business Associate Agreement (Health Insurance Portability and Accountability Act of 1996)

The Z-CAN Provider Manual required physicians to complete reports and forms on a regular basis including:
- Master Patient ID List (submitted separately from forms containing visit encounter information)
- Contraception Supply Orders (including amount remaining in stock)
- Initial Visit Patient Encounter Form
- Return Visit Patient Encounter Form
- Adverse Event Form (submission required within 24 hours of awareness of event)

Lessons Learned: Physician Reimbursement
Reimbursement rates were part of the Physician Agreement . However, project staff still needed to remind physicians about reimbursement rates continuously, in various forms of communication, throughout the project period. The Z-CAN reimbursement rate was commensurate with Medicaid reimbursement rates for IUD and implant insertion and removal in the continental United States. The median reimbursement rates for IUD insertion and removal and implant insertion and removal were used to establish a bundled (combined) reimbursement to ensure that women could have their IUD and implant devices removed when they chose at no cost, even after the Z-CAN program ended.
The Z-CAN reimbursement also covered client-centered contraceptive counseling. It was important to emphasize throughout the project that reimbursement payments included both insertion and removal of LARCs, and that physicians would not receive additional reimbursement at the time of removal.
RESOURCES AVAILABLE:
Your organization may have extensive experience in planning and executing high-quality trainings for staff, providers, administrators, and leaders in the proposed program area. If training is not your specialty, this overview highlights considerations that should be part of your planning.
Comprehensive training for staff is essential, whether the context is public health, clinical care, the development sector, or complex humanitarian emergencies. Regardless of where services will be delivered, training aimed to increase access to contraception should include the following considerations: best practices, local norms, evidence base, cultural context, and baseline capacity of trainees.
- Where should the training take place?
- Will it be centralized, or at individual locations?
- Will the trainers go onsite, or will trainers be virtual?
- How often and when should we schedule training?
- How often does it need to occur?
- What is the best time for the training?
- Time trainings to maximize participation and minimize the burden.
Z-CAN Example: Training
In preparation for Z-CAN trainings, we created clinic toolkits for providers and staff. We held several centralized trainings over the course of two months to train the maximum numbers of providers quickly due to the Zika virus outbreak.
RESOURCE AVAILABLE:

Key points:
- Consider a discussion-based training with minimal lecture instruction or a limited PowerPoint presentation.
- Consider distributing materials to participants for review before training, as in a “flipped-classroom” approach. Advance distribution facilities a discussion-based approach to the pre-reviewed materials.
- Use the Training Plan Template to assist in the planning process.
- What should the training curriculum look like?
- Design a training curriculum that aligns with:
- Gaps identified in the needs assessment
- Recruitment process
- Program goals
- Design the training to accommodate different levels of pre-existing knowledge among trainees.
- Apply adult learning methodology, selecting a training method that fits the content. The training environment is also important.
- Design a training curriculum that aligns with:
- Pre- and post-training knowledge assessments can be very useful to assess the impact of
the training.
- Use assessment after the training to identify remaining knowledge gaps.
- If possible, align these instruments or frequently asked questions with previously validated questions adapted to the local cultural and language contexts.
- Consider piloting these instruments with local health care providers and staff not involved in training.

Lessons Learned: Training
- Include enough family planning experts to supervise hands-on simulation and assess competency of every trainee. Pre-training knowledge assessment can determine how many experts will be needed to supervise hands-on simulation.
- Balance group discussion with didactic/lecture sessions, as much as training size will allow, to assess trainees’ knowledge throughout training.
- adapting and tailoring evidenced-based materials to meet specific project needs during planning and implementation can save time and resources. Using these existing resources allows a program to build on lessons learned from other partners and projects.
- Partners with specific clinical and technical expertise can assist with specific clinical needs such as training and mentoring.
- Client-centered contraception counseling curriculums and hands-on IUD simulators exist, but a project clinical may be able to replicate a training curriculum based on firsthand knowledge and publically available materials and presentations.
RESOURCES AVAILABLE:
Training is only one part of increasing knowledge and capacity to deliver high-quality services. Onsite clinic mentorship and program readiness assessment also are essential to quality assurance.
- Readiness assessment allows the program to evaluate whether a site currently has the capacity to provide the program services to the community.
- Onsite clinic mentorship allows an expert who is well versed in the proposed program
specifications to share knowledge and experience one-on-one with those who will provide
services. Adding onsite mentorship can:
- Enhance new learning
- Strengthen procedural skills
- Acknowledge changes to existing practice
- Create opportunities to discuss additions to the practice scope
- Identify new roles
Knowing that all skills, equipment, and processes are in place builds provider and staff confidence that they can deliver the service. Finally, readiness assessment and onsite mentoring make it clear that the site will support sustainable delivery of the needed program intervention.
RESOURCES AVAILABLE:
- 2.4a Z-CAN Provider- Level Proctoring Checklist
- 2.4b Z-CAN Clinic-Level Proctoring Checklist
- 2.4c Z-CAN Clinic Supply and Equipment List
Z-CAN Example: Mentoring
Before program implementation, a family planning physician and a Z-CAN program staff member conducted an onsite mentorship visit with each provider and clinic to ensure their readiness. This was a time-consuming step as we mentored over 150 physicians, but it was critical to ensure high-quality services would be provided during this rapid implementation and scale-up of a new service across the island during a public health emergency.

Each program will decide what activities require proficiency and how to determine proficiency. Z-CAN physician and clinic readiness criteria included:
- Client-centered counseling through direct observation of care by staff or health care providers
- Understanding and use of contraception eligibility criteria and use of job aides to provide evidence-based care
- Provision of short-acting methods
- Insertion and removal of various LARC methods (direct observation of at least one IUD insertion)
- Direct observation of staff interaction with patients
- Review of data collection, inventory tracking, and billing procedures
- Clinic audit to ensure that supplies, space, equipment, and security were sufficient for Z-CAN program implementation
- Overall clinic readiness to participate in the program
Obtaining products and distribution agreements with local companies, if available, can help build sustainability for programs. Before a project begins, it is imperative to obtain appropriate regulatory approvals from agencies in the location of the project. For example, in the United States, those agencies are the U.S. Food and Drug Administration, Centers for Medicare and Medicaid Services, Department of Health and Human Services, Office of the Inspector General, U.S. Department of Veterans Affairs and the state Departments of Health. These agencies will ensure that the project does not violate the various statutory authorities overseen by the agencies. Local licensed pharmaceutical distributors are aware of target guidelines and how to meet requirements within their facility. Developing communication channels to ensure frequent contact with the distributor is important to discuss par levels (minimum quantity of product that should always be in stock), upcoming orders from manufacturer, upcoming orders to customer, products expiring, and customer inquiries.
Z-CAN Example: Product Procurement and Distribution
To ensure compliance with regulatory guidelines for distribution of pharmaceutical products, CDC Foundation identified and contracted with a local licensed pharmaceutical distributor in Puerto Rico to receive, store, and distribute contraceptive products. Once the agreements with pharmaceutical manufacturers were established, the CDC Foundation team placed orders with the manufacturers to ship contraceptive products to the licensed pharmaceutical distribution warehouse. The CDC Foundation managed the par levels of all contraceptive products in the warehouse except for implants, which were sent directly (drop shipped) from the manufacturer to the physician or clinic, pursuant to manufacturer regulations. The PRDOH issued Z-CAN physicians a "Botiquín" (locked medicine cabinet) waiver to allow onsite stocking of the Z-CAN contraceptive methods to provide same-day services for the duration of the Z-CAN program. The PRDOH required participating physicians and their respective clinic(s) have a valid Botiquín waiver on file.
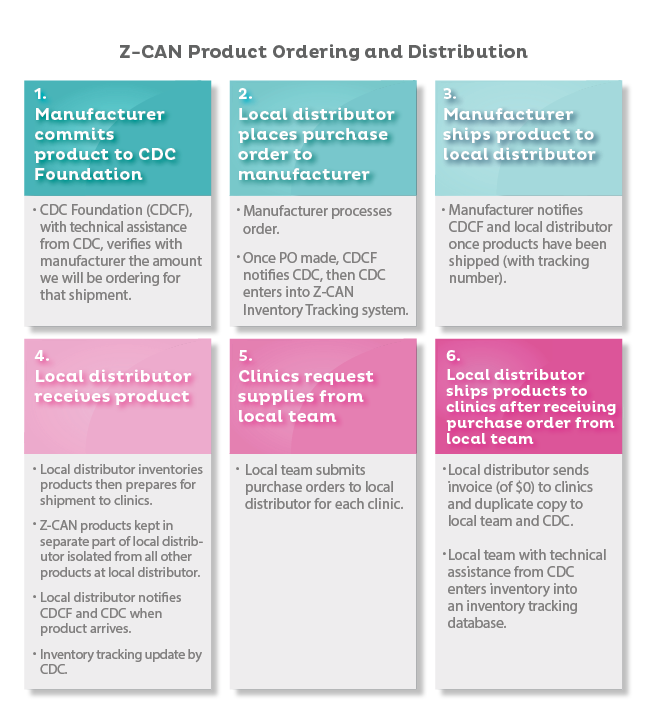
Z-CAN Product Ordering and Distribution
- Manufacturer commits product to CDC Foundation
- CDC Foundation (CDCF), with technical assistance from CDC, verifies with manufacturer the amount we will be ordering for that shipment.
- Local distributor places purchase order to manufacturer
- Manufacturer processes order.
- Once PO made, CDCF notifies CDC, then CDC enters into Z-CAN Inventory Tracking System.
- Manufacturer ships product to local distributor
- Manufacturer notifies CDCF and local distributor once products have been shipped (with tracking number).
- Local distributor receives product
- Local distributor inventories products then prepares for shipment to clinics.
- Z-CAN products kept in separate part of local distributor isolated fro all other products at local distributor.
- Local distributor notifies CDCF and CDC when product arrives.
- Inventory tracking update by CDC.
- Clinics request supplies from local team
- Local team submits purchase orders to local distributor for each clinic.
- Local distributor ships products to clinics after receiving purchase order from
local team
- Local distributor sends invoice (of $0) to clinics and duplicate copy to local team and CDC.
- Local team with technical assistance from CDC enters inventory into an inventory tracking database.
CDC Foundation worked with the pharmaceutical distributor to set up individual accounts for each Z-CAN physician and clinic. Once Z-CAN physician clinics were certified to provide Z-CAN services, the CDC Foundation placed an initial start-up supply order for all respective clinics. Clinic staff initiated subsequent supply orders by submitting a Supply Order form via the Z-CAN e-mail or fax. After weekly reviews to confirm the requested amounts were appropriate, the orders were submitted to the local pharmaceutical distributor to process for shipment.
Lessons Learned: Product Procurement and Distribution
Investing sufficient time and resources is critical for rapid start-up of a contraceptive access program, especially in an emergency response.
- Procurement of Products: If products are shipped outside of the United States or U.S. territory, program staff will need to work with the manufacturer and local distributor to acquire all the appropriate documentation for the country’s customs agency. Fortunately, Z-CAN products did not have to go through U.S. Customs and Border Protection since Puerto Rico is a U.S. territory.
- Track and Trace of Product: It is easier to track products and distribution if each provider or clinic that supplies the service has an individual account. The local pharmaceutical distributor should establish accounts for each individual provider by clinic (if multiple clinics). It is essential for program staff to hold regular conference calls with the local pharmaceutical distributor to discuss par levels, upcoming and pending orders, and any product inquiries.
- Managing Inventory Levels at Clinics: It is important to train clinic staff to manage product par levels. Clinics need to be able to track receipt and provision of all contraceptives provided through the program for reporting and to ensure products are always in stock.
Developing a data collection and management system is essential for gathering information to monitor your program activities. Your team will review data regularly to gauge the program’s effectiveness.
This process also may include tracking patient data and evaluating the impact of the program.
There are many widely available resources that describe basic data management principles and systems. Consult the appropriate personnel at your institution to learn what resources are available to your program and to discuss security issues affecting the collection and storage of data.
The requirements for collecting and storing data containing personally identifiable information (PII) or protected health information (PHI) are stricter than the requirements for storing de-identified data, so consider this factor when designing your data collection instruments. Follow the guidelines in place at your institution for the protection of PII and PHI.
RESOURCE AVAILABLE:
Z-CAN Example: Data Collection and Management
The Z-CAN program developed a data collection and management system to track both patient outcomes and the receipt and provision of all products used by the program.
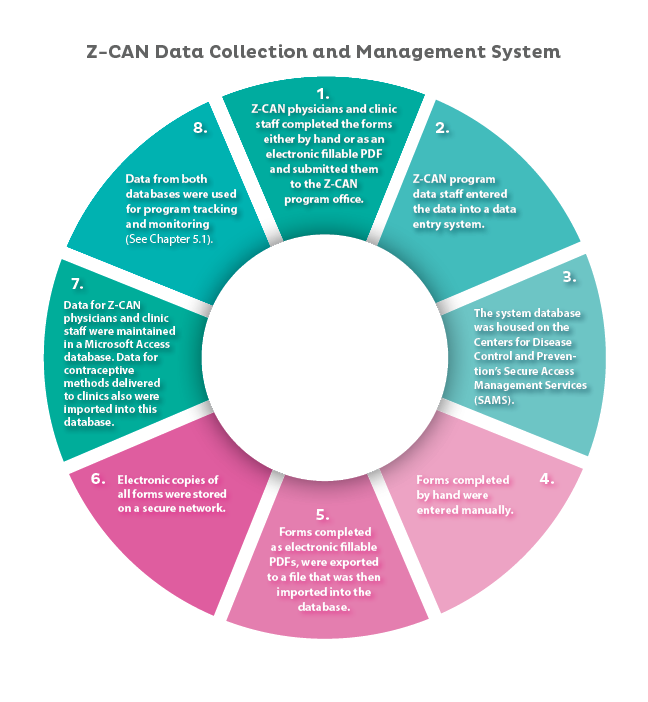
Z-CAN Data Collection and Management System
- Z-CAN physicians and clinic staff completed the forms either by hand or as an electronic fillable PDF and submitted then to the Z-CAN program office.
- Z-CAN program data staff entered the data into a data entry system.
- The system database was housed on the Centers for Disease Control and Prevention’s Secure Access Management Services (SAMS).
- Forms completed by hand were entered manually.
- Forms completed as electronic fillable PDFs, were exported to a file that was then imported into the database.
- Electronic copies of all forms were stored on a secure network.
- Data for Z-CAN physicians and clinic staff were maintained in a Microsoft Access database. Data for contraceptive methods delivered to clinics also were imported into this database.
- Data from both databases were used for program tracking and monitoring (See Chapter 5.1).
Data from both databases were used for program tracking and monitoring (See Section 5.1).